Impax Laboratories, Inc. announced earlier this week that EMVERM (mebendazole) 100 mg chewable tablets are now available for healthcare providers to prescribe to patients in the United States.
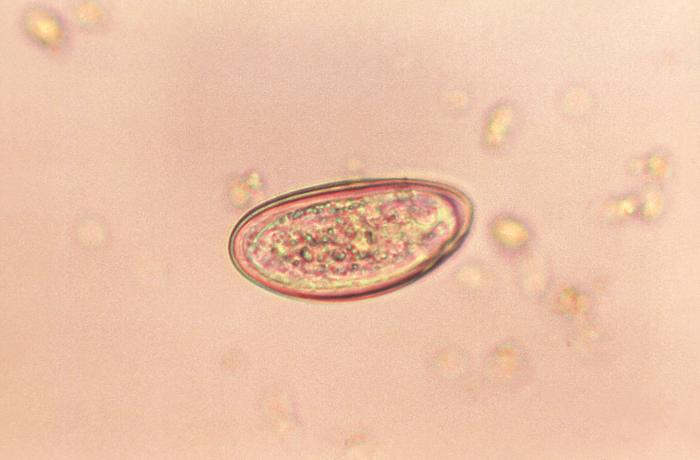
EMVERM is indicated for the treatment of Enterobius vermicularis (pinworm), Trichuris trichiura(whipworm), Ascaris lumbricoides (common roundworm), Ancylostoma duodenale(common hookworm), Necator americanus (American hookworm) in single or mixed infections.
Pinworm is a highly contagious parasite that infects approximately 40 million people in the United States each year. Pinworm infection is three times more common than head lice. EMVERM offers a 95% cure rate in a single 100 mg dose. EMVERM is not for persons who have shown hypersensitivity to the drug.
For additional information, see the Impax Laboratories Press Release
Related:
- UTI treatment: FDA grants Fast Track Designation to investigational antibiotic, CARBAVANCE
- CLL treatment, Venclexta receives FDA approval
- Genital herpes treatment: Genocea Biosciences announce data from Phase II trials



2 thoughts on “Chewable parasite drug, EMVERM, now available for treatment pinworm, others”