By NewsDesk @infectiousdiseasenews
Since the beginning of the year through July 5, the Democratic Republic of the Congo (DRC) has reported a total of 2,591 suspected monkeypox cases and 97 deaths.
This includes 115 cases and four deaths recorded during the week ending July 5.
In 2019, DRC saw 5,288 monkeypox cases, including 107 deaths (CFR: 2%) from 133 health zones in 19 provinces.
The World Health Organization says one major challenge to the current emergency include acquiring the required funding to respond to all the multiple ongoing outbreaks in the country.
In addition to the monkeypox outbreak, the DRC is battling outbreaks of Ebola, measles, malaria, acute respiratory infections, typhoid, COVID-19, cholera, meningitis, diarrhea with dehydration in children under the age of five years, influenza and yellow fever.
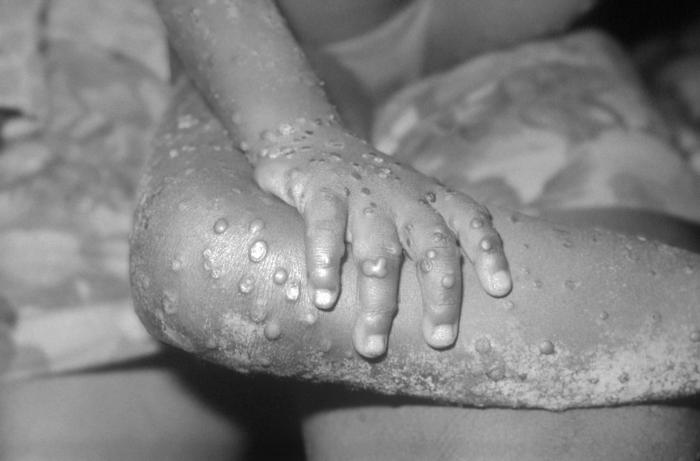
Monkeypox is a rare disease that occurs throughout remote parts of Central and West Africa, often near tropical rainforests. It is spread through contact with the monkeypox virus from an animal or human (alive or dead) or with materials contaminated with the virus.
Symptoms begin with fever, headache, muscle aches, swollen lymph nodes and exhaustion, and is followed by a rash. Patients are usually ill for 2-4 weeks. Monkeypox is fatal in as many as 10% of people who get it.
Got Smallpox? merchandise



Great interview. Would disagree that there is no antiviral available.
TOPXX or Tecovirimat is a smallpox antiviral that was recently FDA approved and is stockpiled in the US. Many of the animal studies used Monkeypox and it was very effective. I’m curious why is isn’t being used in the current outbreak.
Ron, you appear to be very knowledge. I had never heard of Monkeypox until reading this article. I go to South Central Congo about 6 months out the year. Ebola is more of a problem in my area.
Ebola much more deadly for sure. There’s been more human to human transmission transmission of Monkeypox which is concerning. That caught my attention.
Ron, The interview was from Aug 2018
I missed that. The article was new but they threw in an old interview. Thanks.
At the moment, tecovirimat has only been approved as you say, as an emergency preparedness medical countermeasure against smallpox (should the need ever arise). I agree that the manufacturers and FDA should also pursue licensing for monkeypox, which would make it much easier to proceed, rather than require.
There is a reason why tribes would label regions taboo and tell people not to go in those areas. They understood people who did would die and thought the place was curses when in fact it was a place where they could acquire infections. Now with people desperate for land they are going in such regions and bringing out contagions that put the whole world at risk. Before this age of travel deaths would run out in a local confined area but they now can jump around the globe.
At the moment, tecovirimat has only been approved as you say, as an emergency preparedness medical countermeasure against smallpox (should the need ever arise). I agree that the manufacturers and FDA should also pursue licensing for monkeypox, which would make it much easier to proceed, rather than require emergency use authorization.