Argos Therapeutics, Inc., a biopharmaceutical company focused on the development and commercialization of fully personalized immunotherapies for the treatment of cancer and infectious diseases based on the Arcelis® technology platform, today announced that the NIH Division of AIDS (DAIDS) has approved $6.6 million in funding for an investigator-initiated Phase 2a adult eradication study of AGS-004, the company’s investigational fully personalized immunotherapy for HIV.
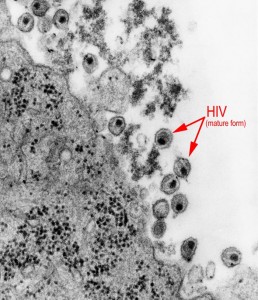
Eliminating the latent virus reservoir has been shown to be an essential goal in efforts to eradicate HIV. Research has shown that histone deacetylase (HDAC) inhibitors can activate cells that are latently infected with HIV, making them more visible to the immune system. One strategy to achieve HIV eradication is known as the “kick and kill” approach where a latency reversing therapy such as an HDAC inhibitor is combined with an immunotherapy to maximize the immune system’s response to the latent reservoir in an effort to eliminate it.
“Previous study results suggest AGS-004 can induce anti-viral memory T-cell responses that are associated with lower persistent viral reservoirs when administered in combination with standard antiretroviral therapy (ART),” said Dr. Charles Nicolette, Argos’ chief scientific officer and vice president of research and development. “The continued support of DAIDS allows us to move forward with stage two of our adult eradication trial, which has already been cleared for initiation by the FDA. We believe that this is the first human clinical trial to test the highly promising ‘kick and kill’ approach to treatment.”
“With this NIH funding, we can now study whether combining AGS-004 treatment with a latent reservoir mobilizer can lead to the elimination of HIV-infected cells,” stated Dr. David Margolis of the University of North Carolina, principal investigator of the AGS-004 Phase 2a adult eradication study. “In stage two of this study, patients on ART will receive the HDAC inhibitor vorinostat in addition to AGS-004.”


I realy need that cure
hey i also get cured by this man please contact him through this mail [email protected]
Am from south Africa…i saw this comment on positive blogs and i will love to tell every body how my status changes to negative, and am now a living witness of it and i think its a shame on me if i don’t share this lovely story with other people infected with this deadly virus…,HIV has been ongoing in my family… i lost both parents to HIV,. and it is so much pain Ive not been able to get over.. as we all know medically there is no solution to it..and medication is very expensive. So someone introduced me to a native medical practitioner in Africa..i had a job there to execute so i took time to check out on him.i showed him all my tests and results.. i was already diagnosed with HIV and it was already taking its towel on me.. i had spent thousands of dollars so i decided to try him out…i was on his dosage for 3 weeks. although i didn’t believe in it, i was just trying it out of frustration… and after 2 weeks, i went for new tests… and you wont believe that 5 different doctors confirmed it that am negative..it was like a dream,,i never believe aids has cure..am now negative,,am a living witness..i don’t know how to thank this man… i just want to help others in any way i can..have joined many forums and have posted this testimonies and a lot of people has mail and called this man on phone and after 2 months they all confirmed negative..BBC news took it live and every.. hope he helps you out.. everybody saw it and its now out in papers and magazines that there’s native cure for HIV and all with the help of this man,,have tried my own parts and all left with you,,if you like take it or not..god knows have tried my best.about 97 people have been confirmed negative through me..and they send mails to thanks me after they have been confirmed negative,,this man is real..don’t miss this chance,,hiv is a deadly virus,,get rid of it now..
FREEHIVCUREHOME@Email,COM or call +2348077309081