Fresh cilantro from the state of Puebla, Mexico has been implicated in annual recurring outbreaks (in 2012, 2013, and 2014) of cyclosporiasis in the United States and two states reporting cases this summer have identified cilantro from this region as a suspect vehicle with respect to separate illness clusters, according to an Food and Drug Administration (FDA) Import Alert published today.
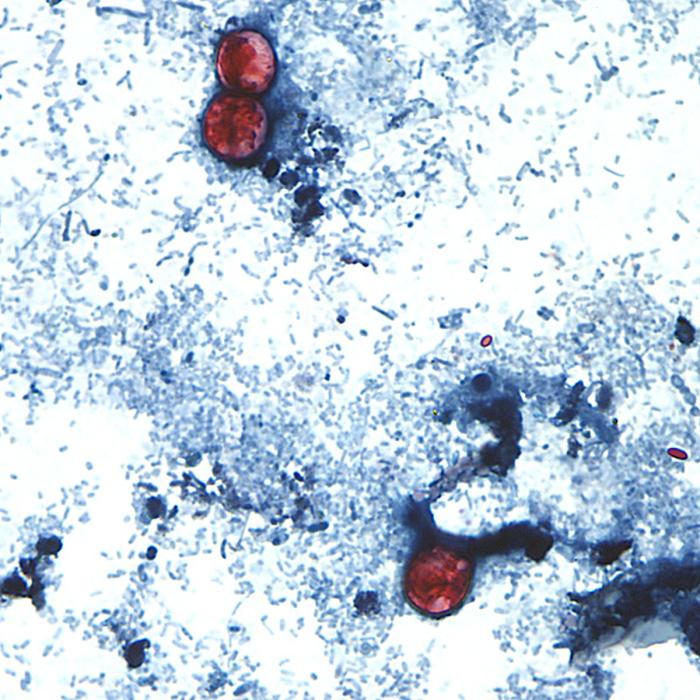
The Texas Department of State Health Services has identified cilantro from the state of Puebla as a suspect vehicle in an ongoing outbreak (as of May 2015). To date in 2015, Texas has reported 205 cyclospora cases statewide.
In Wisconsin, officials have identified cilantro from the state of Puebla, MX as a suspect vehicle for a cluster of illnesses associated with a single restaurant. According to Jan Klawitter with Public Relations with the Wisconsin State Laboratory of Hygiene, the case count is 8 for this outbreak cluster investigation.
FDA believes it is extremely unlikely that these outbreaks of cyclosporiasis are due to isolated contamination events because of their recurring nature, both in the timing with which they occur (typically April to August each year) and the repeated association of illnesses with cilantro from the state of Puebla.
No single supplier (including retail outlets or distribution centers), packing date, shipping date, or lot code can explain all the illnesses.
FDA believes the source of C. cayetanensis contamination is likely attributable to a broader source of contamination. Sources of contamination may include fecal contamination of growing areas, irrigation of fields with water contaminated with sewage, cleaning or cooling produce with contaminated water, and/or poor hygienic practices of workers that harvest and process the produce, and lack of adequate cleaning and sanitizing of equipment that comes in contact with the product.
The FDA said they will detain Mexican cilantro at the border from April to August and won’t allow products from the state of Puebla, Mexico, into the U.S. without inspections and certification.
Related:
Texas cyclospora outbreak linked to fresh cilantro from Mexico in some cases
Cyclospora outbreak in Iowa and Nebraska linked to Taylor Farms de Mexico salad mix: FDA
Cyclospora outbreak expands to 19 states, Taylor Farms de Mexico suspends production of salad mix


3 thoughts on “Puebla, Mexico fresh cilantro ‘suspect’ in Texas cyclospora outbreak and Wisconsin cluster”