By NewsDesk @bactiman63
Congressional Valley Fever Task Force Co-Chair and Congressman Kevin McCarthy, along with Task Force Co-Chair and Congressman David Schweikert (AZ-06), Congresswoman Karen Bass (CA-37), and Congressman Tom O’Halleran (AZ-01) introduced a bipartisan bill, the Finding Orphan-disease Remedies with Antifungal Research and Development (FORWARD) Act of 2021 (H.R. 5566), to combat Valley Fever. Specifically, this bill would work to address challenges related to detecting, treating, and ultimately eliminating Valley Fever by incentivizing antifungal therapeutics and vaccine development.
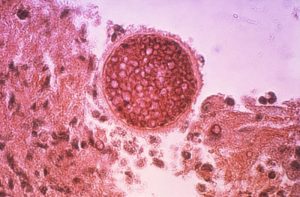
McCarthy released the following statement:
“For years, our community has worked to tackle Valley Fever head on. And though we have made progress, we have never forgotten the critical need to find effective treatments, a cure, and ultimately a vaccine. This week served as an important reminder of that need – high wind advisories in Kern County caused Valley Fever to be a greater-than-normal threat in our community given that strong winds are notorious for kicking up the fungal spores that cause Valley Fever, making cures and a vaccine all the more important.
“As we continue to recover from a global pandemic resulting from viral respiratory illness COVID-19, we must refocus our efforts to better address fungal lung diseases, such as Valley Fever, which can complicate COVID-19 response efforts. The FORWARD Act takes a robust approach to combating this disease by focusing on short-term, medium-term, and long-term solutions to develop new treatments, therapies, and a vaccine. I am thankful to Congressman Schweikert for his leadership on this issue over the years, and I look forward to continue working with him and our colleagues in Congress to finally stamp out Valley Fever.”
Schweikert offered the following statement:
“Arizonans continue to be disproportionately affected by Valley Fever, an endemic fungal disease that impacts broad areas across America’s Southwest. That is why I am extremely proud to join my Congressional Valley Fever Task Force Co-Chair and Congressman Kevin McCarthy, alongside my colleagues, on introduction of the Finding Orphan-disease Remedies with Antifungal Research and Development (FORWARD) Act of 2021. This bipartisan legislation will provide additional funding, raise awareness, and accelerate the delivery of medical breakthroughs that medical professionals across the country are working tirelessly to produce. I look forward to continuing my work alongside the leadership of Congressman McCarthy who fights tirelessly against Valley Fever.”
Leading Valley Fever researchers, health professionals, and patient advocates support the FORWARD Act:
“The FORWARD act brings much needed attention and support to the endemic fungi, such as those that cause Valley Fever, a public health disease so important to residents and visitors to the desert southwest. This year in Arizona, our data shows that for every four new diagnoses of COVID-19, an additional person with the same symptoms has Valley Fever. While COVID-19 is very important now, Valley fever is an endemic problem year in and year out.” –Dr. John Galgiani, Director of the Valley Fever Center for Excellence at the University of Arizona College of Medicine-Tucson
The FORWARD Act is designed to advance sustained efforts to combat Valley Fever in the short, medium, and long-term. Specifically, the FORWARD Act would in the:
- Short-Term: Immediately support and prioritize basic research for Valley Fever and other fungal diseases, authorize $20M annually through FY26 for National Institutes of Health (NIH) to enter into grants or contracts for vaccine/drug development, and create a federal working group to coordinate research efforts on fungal diseases;
- Medium-Term: Streamline the U.S. Food and Drug Administration (FDA) approval process to get new antifungal diagnostics, treatments, and vaccines approved for use in humans, as well as expand and codify at current funding levels a successful program, renamed the Combating Antimicrobial Resistance Biopharmaceutical Accelerator (CARB-X) Program in this legislation, to promote public-private partnerships to develop both antibacterial and antifungal diagnostics, treatments, and vaccines; and,
- Long-Term: Encourage the development of a Valley Fever vaccine by extending expedited approval pathways for antifungal vaccines and directs the FDA to include Valley Fever in its priority review voucher program to further incentivize the development of new treatments, cures, and vaccines.
To stay up to date on Congressional efforts to address Valley Fever, please visit the Congressional Valley Fever Task Force website.


One thought on “Valley fever bipartisan bill, FORWARD Act, introduced”