Cambridge, Mass. biopharmaceutical company, Genocea Biosciences, Inc., announced this week positive 12 month efficacy data from its Phase 2 dose optimization trial evaluating GEN-003 for the treatment of genital herpes.
GEN-003 demonstrated sustained and statistically significant reductions compared to baseline in the rate of viral shedding 12 months after dosing across multiple dose groups as well as sustained efficacy at multiple dose levels across secondary endpoints measuring the impact on clinical disease. GEN-003 was safe and well tolerated by patients, with no serious adverse events related to the vaccine in the trial.
“We are very pleased with these data, which show that GEN-003 has strong and durable effects on both HSV-2 viral activity and genital herpes clinical disease, supporting our belief that GEN-003 could become a cornerstone treatment for patients affected by this serious disease. Specifically, a single course of treatment of GEN-003 may offer benefits similar to a full year of daily administration of oral antivirals – but with greatly improved convenience,” said Chip Clark, president and chief executive officer of Genocea. “We anticipate reporting virologic efficacy data for GEN-003 from our recently-initiated Phase 2b study in the third quarter of 2016, clinical efficacy data at 6 months post dosing around the end of 2016 and conducting our end of Phase 2 meeting with the FDA in the first quarter of 2017.”
“These 12 month data highlight the potential of GEN-003 to significantly enhance the genital herpes treatment landscape,” said Lori A. Panther, M.D., MPH, infectious diseases specialist at Beth Israel Deaconess Medical Center and Assistant Professor of Medicine at Harvard Medical School. “Because of the physical and psychological impact of this disease, both patients and treating physicians would be eager to use an effective treatment that more conveniently improves control of outbreaks. The reduction in viral shedding, which is thought to cause the epidemic spread of genital herpes, is also encouraging.”
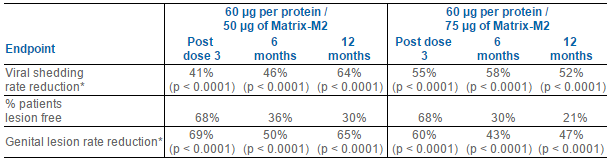
Genocea has already advanced the two most promising doses, 60 µg per protein combined with either 50 or 75 µg of Matrix-M2TM adjuvant, from this Phase 2 dose optimization study into an ongoing Phase 2b efficacy trial. The efficacy of GEN-003 at these two dose levels over the course of the Phase 2 dose optimization trial is as follows:
Inducing a T cell response against HSV-2 is critical to treating the clinical symptoms of disease and controlling transmission of the infection. GEN-003 is a first-in-class T-cell directed immunotherapy designed to elicit both a T cell and B cell (antibody) immune response. The immunotherapy was designed using Genocea’s ATLAS™ platform, which profiles the comprehensive spectrum of actual T cell responses mounted by humans in response to disease, to identify antigen targets that drive T cell response. GEN-003 includes the antigens ICP4 and gD2 along with Matrix-M2TM adjuvant, which Genocea licenses from Novavax, Inc. For more information about GEN-003, please visit http://www.genocea.com/platform-pipeline/pipeline/gen003-for-genital-herpes/.
Genital Herpes affects more than 400 million people worldwide and causes recurrent, painful genital lesions. It can be transmitted to sexual partners, even when the disease is asymptomatic. Current genital herpes therapies only partially control clinical symptoms and viral shedding, a process which drives disease transmission. Incomplete control of genital lesions and transmission risk, expense and the perceived inconvenience of taking a daily medication are hurdles for long-term disease management. Immunity through T cells is believed to be particularly critical to the control and possible prevention of genital herpes infections.
Related:


Even though 90 percent of people have some form of herpes, the virus is heavily stigmatized. But the transition from “itchy annoyance” to shameful secret may not have been coincidental.
TESTIMONY OF HOW I GOT CURED FOR MY GENITAL HERBS SIMPLES .VIRUS
Believe me, I tried everything including BHT, Lysine, acyclovir, creams,
lotions, toothpaste, zorivax 5\% cream and pills too, bleach, (I would dab it on with a q-tip), ice, anything and everything.As if the sores weren’t enough, there was the horrid pain of neuralgia, which I experienced on every part of my body, (you can’t imagine how painful it gets when it’s on your face!). . When I took a shower, even though the water temperature was normal, it would feel like hot drops of grease or cold drops of sleet.On one faithful day i read a testimony on internet of a lady who was cure by HERBALIST Called doctor Ekpen herbs,so i contacted the same herbalist who send herbal cure to me through FEDEX company and i take the herbs for 1 week and i was fully cure after i got tested by my Doctor.Youcan also contact this herbalist via; Whatsappnumber +2348102454875 orEmail doctorekpen222@gmail.comURINARY TRACT INFECTION,HERPES 1& 2 CANCER, LOVE SPELLBARENESS/INFERTILITY ASTHMA… HIV/AIDS
…HE IS THE ONLY HERBALIST THAT HAVE CURE FOR ORAL AND GENITAL HERPES,TRY
Been diagnosed of herpes is just like been through hell but everything is posible. So amazing and i want to testify of the good work done by dr.Osas a very strong and powerful spell caster. dr.Osas has proved science and scientists wrong for believing and thinking that there is no cure for Herpes Virus but to be sincere this dr.Osas is so real and powerful. I never thought that this cure will work but this GREAT MAN has proved it to me and it has worked and still working perfectly. My Name is James Benand and i was diagnosed of the deadly disease called Herpes April 2016 and i thought that, this will be the end and there will be no hope. Speaking to anyone was always a problem because i was so worried and always in the state of unrest in fact i thought it was over until one blessed day when i was searching for help here on the internet, i came across a testimony about dr.Osas on how he helped a lady who was also infected with Herpes Virus, how he has also helped many others to get their loved ones back, and how he has also help others to cure HIV. At first i thought it was all a lie but i decided to give it a try since there was no option and, when i contacted this MAN he laughed and told me that, that is too small for him to handle but i was still in doubt, and he gave me a form to fill which I did, then he called me and told me that his gods required some items in which he will use in preparing the cure for me. He bought the items and prepared a cure for me, and instructed me on what to do and i OBEYED him completely as he told me. Then he sent a parcel to me through the courier delivery service. it was a holy water and a pure holy oil as described by dr.Osas. I took the holy water and the holy oil and after taking it, he told me to go for check up but I was still in doubt and was so afraid to go for the test but i took a bold step and believed that it is well, and i believed all what he has done and all that he has told me. So i went to the hospital for the test it was so amazing, awesome and exciting what seems to be impossible became possible, the VIRUS disappeared completely from my system quickly I called him and told him what happened he congratulated me and today i am a happy person again the way things use to be. Don’t you think that this testimony is worth sharing? I promise to tell the world about him and i will keep sharing this testimony till the end because many people has died of herpes and i want many people who believed that it is over to know that EVERYTHING IS POSSIBLE AND I WANT THEM TO GET HELP FROM THIS SPELL CASTER by contacting him on his email at dr.osascuringhome@gmail.com
All these post about HERBAL MEDICINE and spell casters are scammers and fake trying to collect money all in the name of HERPES cure and when you contact them, they will be asking for your name, pictures, country, phone number, occupation and when you have given them all the information, they will be as king for money. I was once suffering from GENETIC HERPES and i spent a lot of money going from one herbal doctor to another but they all scam and take my money away until i came in contact with this powerful one called Dr.Osas who cured me from Herpes with his herbal medication , after doctor Osas has finished preparing the herbal medicine, he asked me to send him my residential address, so that he will send the herbal medicine to via D.H.L so i did. 2days later, i received the herbal medicine and i had to follow the instructions he gave to me on how i will apply it. 2weeks later i went to hospital for test and according to the test done by the Medical doctor Desmond, it stated that i was cured.
So viewers be wise and beware of fake spell casters and fake herbal doctors, they are all over the internet trying to steal from poor people. I will drop you the email of this powerful herbal doctor just in case you wish to contact him for help. dr.osascuringhome@gmail.com.
Dr OSAS CAN AS WELL CURE THE FOLLOWING DISEASES:-
1. HERPES
2. LASSA FEVER
3. GONORRHEA
4. HIV/AIDS
5. LOW SPERM COUNT
6. MENOPAUSE DISEASE
7. EPILEPSY
8. ASEPSIS
9. CANCER
10. ANXIETY DEPRESSION
11. PREGNANCY PROBLEM
12. SHORT SIGHTEDNESS PROBLEM
14. Male menopause
15. Menopause – male
16. Menopause – peri
17. Menstruation problems
18. Mercury Poisoning
19. Migraine
20. Miscarriage
21. Mites (demodex mites)
22. Mites (scabies mites)
23. Motion sickness
24. Mouth ulcer
25. MRSA
26. Multiple sclerosis
27. Muscle cramps
28. Myodesopsia
29. Stroke
All these post about HERBAL MEDICINE and spell casters are scammers and fake trying to collect money all in the name of HERPES cure and when you contact them, they will be asking for your name, pictures, country, phone number, occupation and when you have given them all the information, they will be as king for money. I was once suffering from GENETIC HERPES and i spent a lot of money going from one herbal doctor to another but they all scam and took my money away until i came in contact with this powerful one called Dr.Cletus who cured me from Herpes with his herbal medication , after Dr.Cletus has finished preparing the herbal medicine, he asked me to send him my residential address, so that he will send the herbal medicine to via D.H.L so i did. 2days later, i received the herbal medicine and i had to follow the instructions he gave to me on how i will apply it. 2weeks later i went to hospital for test and according to the test done by the Medical doctor Desmond, it stated that i was cured.
So viewers be wise and beware of fake spell casters and fake herbal doctors, they are all over the internet trying to steal from poor people. I will drop you the email of this powerful herbal doctor just in case you wish to contact him for help. Dr.Cletuscuringhome@gmail.com.
Dr.Cletus CAN AS WELL CURE THE FOLLOWING DISEASES:-
1. HERPES
2. LASSA FEVER
3. GONORRHEA
4. HIV/AIDS
5. LOW SPERM COUNT
6. MENOPAUSE DISEASE
7. EPILEPSY
8. ASEPSIS
9. CANCER
10. ANXIETY DEPRESSION
11. PREGNANCY PROBLEM
12. SHORT SIGHTEDNESS PROBLEM
14. Male menopause
15. Menopause – male
16. Menopause – peri
17. Menstruation problems
18. Mercury Poisoning
19. Migraine
20. Miscarriage
21. Mites (demodex mites)
22. Mites (scabies mites)
23. Motion sickness
24. Mouth ulcer
25. MRSA
26. Multiple sclerosis
27. Muscle cramps
28. Myodesopsia
29. Stroke
PLEASE DO NOT IGNORE ENSURE TO READ TO HELP PEOPLE WITH HIV AND HERPES VIRUS CURE ..Hello all viewer online,,,, HERPES AND HIV has cure
Do anyone still doubt natural herbal medicine ? I’ve seen the great importance
and the great work of natural herbs and the wonderful work they have done in people’s lives. I wonder why people still spend their money on surgery, injections and drugs that can not cure you completely,. Natural herbs can cure all kinds of illness including herpes, diabetics, asthma, HIV, hepatitis, etc. I’ve seen it with my own eyes. I was cured of HIV virus and my aunt and her husband were cured of herpes virus and tested negative after usage of the herbal medicine an i was also cured of hiv and tested hiv negative after usage of the herbal medicine by Dr. MOHAMMED who uses natural herbs to cure different kind of illness. Even Dr. Sebi prove to the whole world that natural herbs can cure all diseases and he cured countless of people using natural herbs. I know is hard to believe but am a living testimony. There is no harm in trying herbal medicine, the herbal medicine was sent to me through DHL delivering company,,, this is not scam, i am a living witness of this, please believe me and save your life and other people’s life who victimize of the virus or any illness . Contact or email Dr.Mohammed on: herbalcure12@gmail.com (phone call /what-sap no): +2349036036397
I am so grateful to the great dr Emuze for curing my herpes disease for the past 7yrs i was having this infection that made me look so horrible, since i have been having this disease i have been in a complete agony weeping everyday, i have gone to several places trying to get a cure but nothing was change, i had to search for solution in the internet, luckily i came across this testimony of how Dr Emuze cured a lady from this terrible affection so i decided to contact him through his email address:(emuzeherbalcure@gmail.com) i told him exactly how i was feeling and he assure me that he will help me to get cured from my disease, i believed him and obeyed all his instruction that he gave to me, afterwards he prepare a cure for me and when i received it and started using it just as he has promised me, i was cured totally within 2 weeks of usage. contact Dr Emuze today on his email to get the herbs from him; emuzeherbalcure@gmail.com
It’s a pleasure for me to write this testimony about how i got my Genital Herpes cured a month ago. i have been reading so many comments of some people who were cured from various diseases by Dr .Hazim, but i never believed them. I was hurt and depressed so I was too curious and wanted to try Dr. Hazim , then i contacted him through his email when i contact him, he assured me 100% that he will heal me, i pleaded with him to help me out. My treatment was a great success, he healed me just as he promised. he sent me his medication and ask me to go for check up after 21 days of taking the medication. i agreed with him i took this medication and went for check up after 21 days of the treatment, To my greatest surprise my result came out negative after the treatment, I’m very happy that i was cured and been healthy again. I waited for 3 weeks to be very sure if I was completely healed before writing this testimony. I did another blood test one week ago and it was still Herpes negative. so i guess its time i recommend anyone going through Herpes, HSV-1 or HSV-2, HIV, HPV, Hepatitis B, Diabetes, Cancer to contact this great man through his Email usmandrhazim@gmail.com OR add him up on whatsApp +2349058026857
Thanks Doctor Hazim Usman
HOW I WAS CURE FROM HERPES WITHIN TWO WEEKS WITH THE HELP OF DR EHIGIE HERBAL MEDICINE. HIS EMAIL IS EHIGIEHERBALCURE@GMAIL.COM
I am so Happy to be writing this article in here, i am here to explore blogs forum about the wonderful and most safe cure for HERPES SIMPLEX VIRUS . I was positive to the deadly Virus called HERPES and i lost hope completely because i was rejected even by my closet friends. i searched online to know and inquire about cure for HERPES and i saw testimony about Dr Ehigie online on how he cured so many persons from Herpes Disease so i decided to contact the great herbalist because i know that nature has the power to heal everything. i contacted him to know how he can help me and he told me never to worry that he will help me with the natural herbs from God! after 2 days of contacting him, he told me that the cure has been ready and he sent it to me via UPS and it got to me after 5days! i used the medicine as he instructed me (MORNING and EVENING) and i was cured! its really like a dream but i’m so happy! that’s the reason i decided to also add more comment of Him so that more people can be saved just like me! and if you need his help,contact his Email: ehigieherbalcure@gmail.com
He also have the herb to cure difference cure for any sickness
(1) HERPES,
(2) DIABETES,
(3) HIV&AIDS,
(4) URINARY TRACT INFECTION,
(5) HEPATITIS B,
(6) IMPOTENCE,
(7) BARENESS/INFERTILITY
(8) DIARRHEA
(9) ASTHMA…
kindly email DR EHIGIE for your own cure if you have any disease listed above via email: ehigieherbalcure@gmail.com
All thanks belong to Dr Osato, the great herbal man that cured me from HERPES. I got diagnosed 7 months ago and I was desperately looking for a possible way to get this virus out of my body because I believe there is a cure somewhere. I keep searching until i saw people’s testimony about Dr Osato recommendation online for how he has being curing HERPES with his herbs and i emailed him and tell him my problem and he prepare my cure and send it to me through UPS and gave me instructions on how to use the herbal medicine and behold after usage i went to the hospital for checkup and the result was Negative and the symptoms of herpes was completely gone from my body. You can contact Dr Osato to get your herbs too and cure yourself from this horrible virus. His email is osatoherbalcure@gmail. com or through WhatsApp 2347051705853. His website is osatoherbalcure.wordpress.com. Dr Osato cures so many different types of diseases/viruses with his herbs such as HERPES, HIV/AIDS, CANCER of all kinds, HSV 1&2, DIABETES, HPV and so many more. I want to thank God for using Dr Osato to cure me from genital herpes.
Hi Friends I’m so glad writing this article today to tell the world how I got cured from herpes virus. I was diagnosed with HSV-1&2 for two years before Dr Excel cured me with his herbal medication, I contacted him through his website and we end up talking on the phone and then he prepared and sent me the meds which I drink for almost two weeks as he instructed and the virus were gone completely. if you are going through same situation you can also contact him on his website Excelherbalcure.com or you can add him on WhatsApp +1 [438] 792-9488